Facts and Information About Periodic Table for Kids
Mention chemistry, and most people think of a complicated science beyond the grasp of a normal human being to comprehend. If the basics of chemistry are depicted to let students apply them to the world, it becomes a simpler subject. Chemistry is not just restricted to labs, experiments, beakers, and test tubes. Chemistry exists everywhere. Everything in this world comprises elements that are represented in some form. Most of these elements, 118 to be precise, are compressed in the periodic table. Learning more about these elements could make chemistry an exciting subject to learn.
Your adolescent or teenager would be introduced to the periodic table in their chemistry lessons during Grade 7 or Grade 8 at school. At first, the periodic table may seem intimidating and difficult to understand. But the periodic table is a simple arrangement of elements in an understandable manner.
The periodic table is a way of listing the elements on Earth. The structure of their atoms lists elements in the periodic table. This includes the number of protons they have and the number of electrons they have in the outer shell. The elements are listed in the atomic number from left to right and top to bottom, which is the number of protons in each atom.
The periodic table places the elements in a sequential, logical, and easy-to-understand manner based on their physical and chemical characteristics. The elements group in the periodic table is listed on their atomic number, which is the most common factor for categorizing and placement on the table.
Chemical elements that are the essential substances that make matter are arranged in the periodic table. The abbreviations for elements in the periodic table make it easier to read and comprehend. Each element has a peculiar feature known as the atomic number.
What is The Periodic Table?
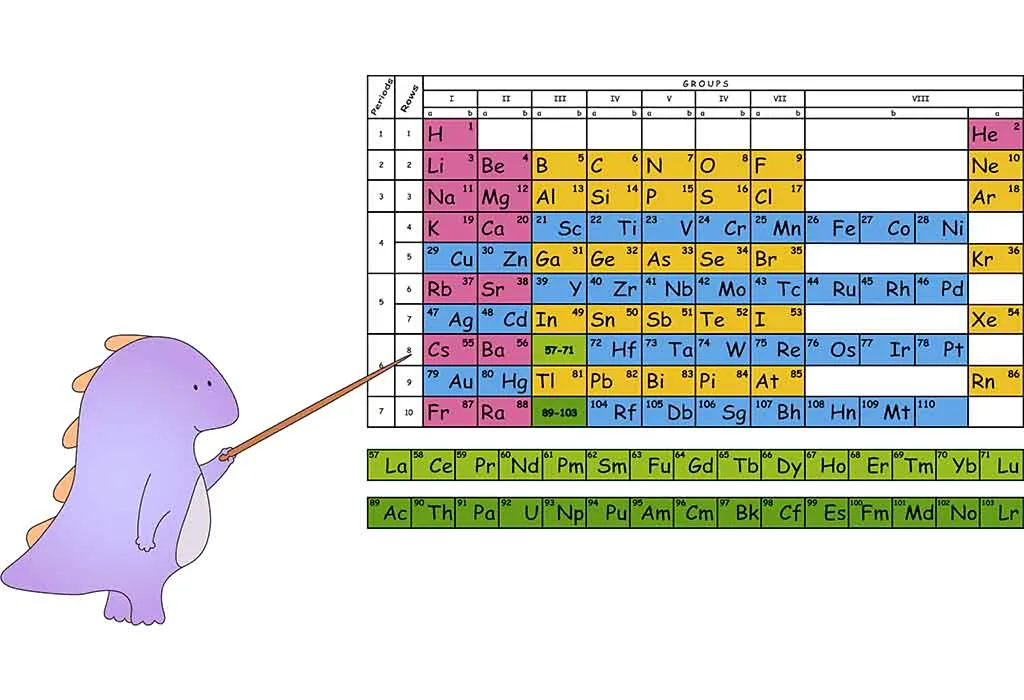
The periodic table is a table that places chemical elements that are essential substances that make up matter in rows and columns. It has 18 columns called groups and seven rows called periods. The name “ periodic table” comes from the seven rows called periods. Each element is placed alongside other elements that share the same characteristics, like noble gas and non-metal. These elements are part of the same group. Elements in the same group share similar characteristics and also react the same way with other elements.
Every chemical element has a peculiar feature called the atomic number. The number comes from the number of tiny particles, called protons, in each atom of the element. In the rows, the elements are placed in order of their atomic number. The columns identify groups of elements with similar chemical properties. E.g., certain metals will be in one column, and certain gases will be in another. The periodic table helps kids learn about elements and their properties.
Where Does The Periodic Table Get Its Name From?
The name originates because the elements are arranged in cycles or periods. Each horizontal row (elements arranged based on the atomic number) is a period, and columns have elements with similar properties. There are seven periods in total, and the first one is short with only two elements- helium and hydrogen. The sixth period has 32 elements. In each period, the element on the left has 1 electron in the outer shell, and the right has an entire shell with the most elements.
Groups are the columns of the periodic table. With 18 columns or groups, each group has different properties. E.g., alkali metals align on the left-most column as they have 1 electron in their outer shell and are reactive.
Who Invented The Periodic Table?
The periodic table was first designed in 1869 by a Russian scientist called Dmitri Mendeleev. Worldwide it was adopted as a new way to represent, classify and organize the elements. It became easy to read and made it easy to teach chemistry with a visual aid.
Up until then, scientists did not know the atomic numbers of elements. They did not even know that each element had its atomic weight. Mendeleev was the one who noticed the relationship between atomic weights and the different properties of elements. When the elements were arranged according to their atomic weight, they were grouped according to shared properties.
The first version created by Mendeleev looked very different, and he revised it several times. As scientists became more aware of elements, the table has been edited several times. The current table which is in use has been the one designed since the mid-1900s.
How Can Kids Read The Periodic Table?
How to explain the periodic table to a child? There are several ways in which the elements in the periodic table are represented. An element could be a part of a row or a column with further sub-classifications in rows, columns, and tables. Interpretations can be different and based on what one is looking for. Below are some ways in which kids could read the periodic table:
1. Fundamental Division
The periodic table has fundamental attributes:
- The seven rows or periods have elements with the same physical properties like conductivity and malleability. Two rows of elements from the atomic number 57-71 (lanthanides) and 89-103 (actinides) are grouped below the main table.
- There are 18 columns or groups where the elements share similar atomic structures and characteristics like valency etc.
2. Sub-groups
One can also categorize elements in the periodic table into three basic types or sub-groups.
- Metals: Elements that share the same characteristic in terms of flexibility, conductivity, and melting point.
- Non-metals: These elements are non-conductors or poor conductors of heat and electricity. These elements are not flexible and lack the characteristic shine or luster of metals.
- Metalloids: These are also called semi-metals which have characteristics between metals and non-metals. Metalloids could be shiny like metals or poor conductors like non-metals. Some metalloids are flexible, while some are not.
3. Column Groups
Elements can be read as part of a group where elements share similar characteristics. Below are the columns:
- Column 1: Hydrogen and alkali metals
- Column 2: Alkaline earth metals
- Column 3 -12: Transition metals
- Column 13-16: Post transition metals, non-metals, and metalloids
- Column 17: Halogens
- Column 18: Noble gases
- Columns 3-16: Consist of lanthanides that are rare-earth elements and actinides. The properties of these elements are different from the rest, which is why they are placed at the bottom of the periodic table.
4. Atomic Number, Atomic Mass, and Atomic Weight
Elements in the periodic table can also be classified according to atomic number, mass, and weight.
- Atomic Number: It is the number of protons ( particles positively charged) that are present in the nucleus of the atom in an element. The letter Z represents it.
- Atomic Mass: This means the number of protons and neutrons ( particles charged neutrally) present in the nucleus of the atom of an element. It is represented by the letter A.
- Atomic Weight: Elements could have multiple isotopes that are different forms of the element with similar atomic numbers ( same number of protons) with a different number of neutrons. Atomic weight is an average of atomic masses of all isotopes of the element.
- Based on the atomic number, atomic mass, and atomic weight, one can observe, interpret and characterize the elements:
- Each element will contain one more proton than the element before it. For example, Helium (He) has an atomic number of 2, one more than the previous element Hydrogen (H). After that, the atomic number (number of protons) will progressively increase as you go further down the table.
- As the number of protons increases, the atomic mass and weight increase as you move down a period or across a group, with some exceptions.
- Groups of elements have the same color scheme and share many physical properties.
- Few element groups could show changes in physical properties as their atomic masses change. For example, noble gases tend to become denser as the atomic mass increases.
- The atomic number( number of protons) of an element remains unchanged. However, there is a possibility of several undiscovered isotopes of the element. The atomic weight could change; therefore, the one mentioned on the periodic table is considered provisional.
Other Facts About Periodic Table For Children
A fascinating tool to classify and categorize elements, the periodic table also has interesting facts. Below are some fun facts about the periodic table for children that they would love to know:
- The periodic table was designed to accommodate elements that are yet to be discovered. New elements could be categorized and positioned in the periodic table. Many elements in the modern periodic table were never a part of the original periodic table designed in 1869.
- The periodic table has several elements, and the symbols of the elements contain almost every letter in the alphabet, barring the letter “J.” New elements are constantly being added, and there is a chance the letter “J” might be assigned to an element as a symbol.
- There are 118 elements in the modern periodic table as of 2021. Ninety-four of the elements occur naturally and are found on Earth. Twenty-four elements that are man-made are called synthetic elements.
- Nihoium, Moscovium, Tennessine, and Oganesson were the most recently added elements in 2016. This was reviewed for five months by the International Union of Pure and Applied Chemistry (IUPAC).
- Carbon is unique and is known to form up to 10 million different compounds. Carbon is essential for the very existence of life.
- Francium is one of the rarest elements on earth. There are no more than a few ounces of it on Earth at any given point in time.
- Argentina is named after the element silver, represented by the symbol (Ag), which is Argentum in Latin.
- Although there is Helium on Earth, it was discovered first by observing the sun.
The periodic table can be regarded as a method of organizing the elements that comprise and make up matter in the order of their protons. It is an essential tool while learning chemistry as a subject. Children need to know about the elements and apply a lot of that while working in chemistry. It is a beautiful idea to get kids to learn the periodic table elements, which will make learning several chemistry topics easier to understand and grasp.
Also Read:
Magnet Facts for Children
Static Electricity Facts for Children
States of Matter Facts for Children
Was This Article Helpful?
Parenting is a huge responsibility, for you as a caregiver, but also for us as a parenting content platform. We understand that and take our responsibility of creating credible content seriously. FirstCry Parenting articles are written and published only after extensive research using factually sound references to deliver quality content that is accurate, validated by experts, and completely reliable. To understand how we go about creating content that is credible, read our editorial policy here.













.svg)